
Metals in Chemistry In chemistry, metals are defined by the properties such as. 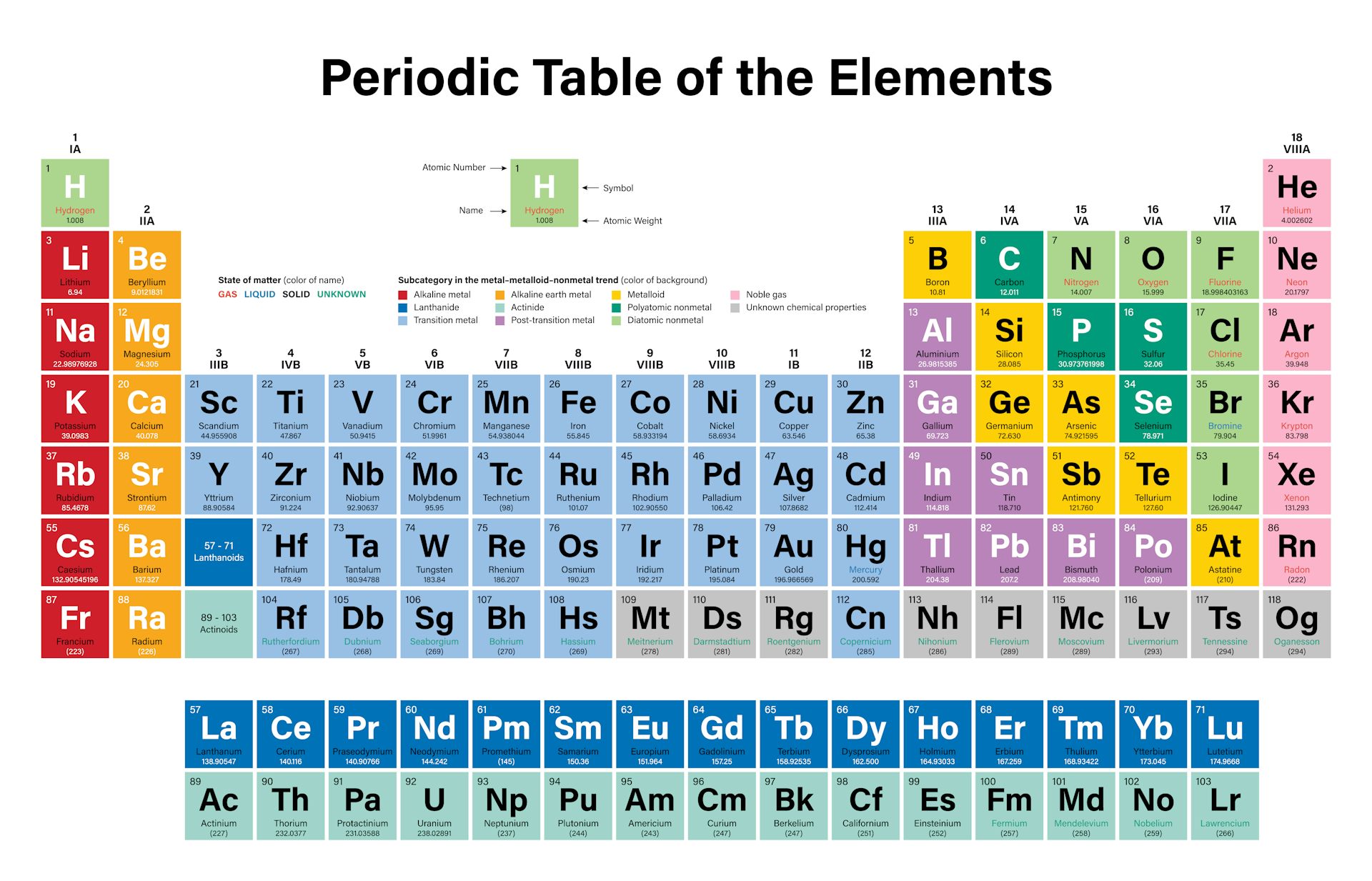
The elements will be considered by group after an introductory section that reviews several concepts that help to make sense of periodic trends in the descriptive chemistry of these elements. Any of a large group of chemical elements, including iron, gold, copper, lead, and magnesium, that readily become cations and form ionic bonds, having. Names of any five metals present in the periodic table are lithium, sodium, zinc, gold, and gallium. Metallic elements can be categorized into different groups in the periodic table alkali metals, alkaline earth metals, transition metals, post-transition metals, lanthanides, and actinides. The alkali metals react vigorously with water, forming hydrogen gas and their. These are (except for hydrogen) soft, shiny, low-melting, highly reactive metals, which tarnish when exposed to air. Chemical elements are placed on the right side of the periodic table, where they are arranged based on their atomic structure. The alkali metals are soft, shiny when freshly cut, have low density, low melting and boiling points, are good conductors of heat and electricity, form white compounds, and are very reactive. Chlorine atoms bridge Al centers in aluminum chloride dimer at left while the structure of pentaborane nonahydride shown at right includes four B-H-B bridge bonds arranged around the open face of a cluster of five B atoms held together by cluster bonds. The alkali metals are the elements found in group 1 of the periodic table.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
